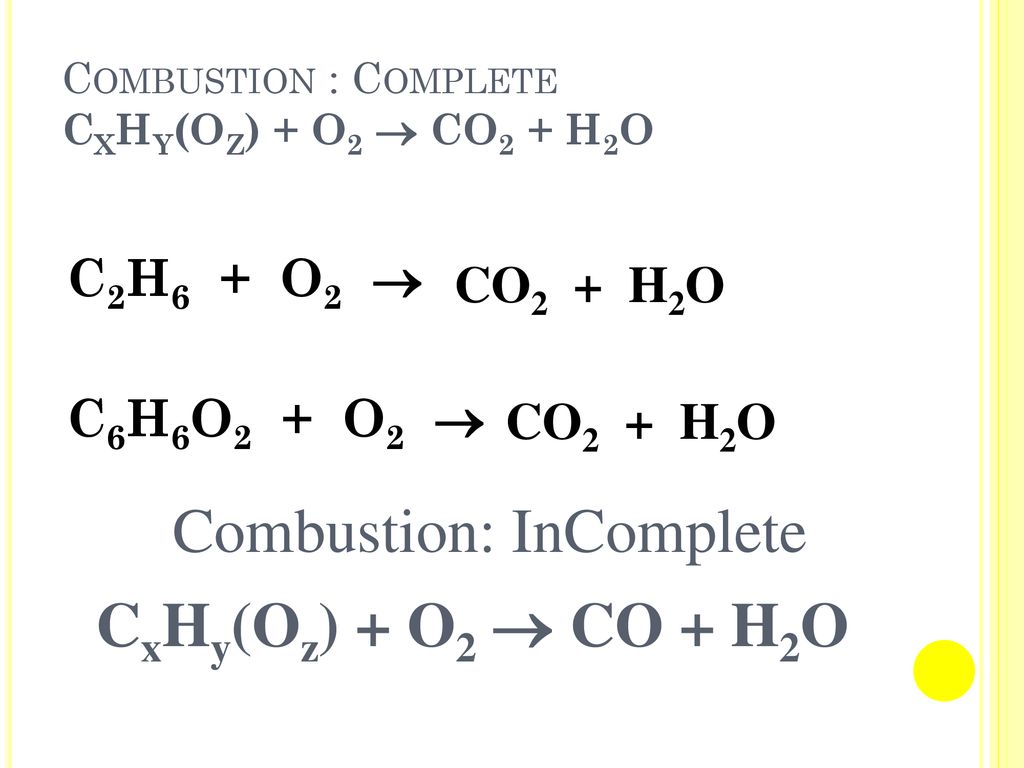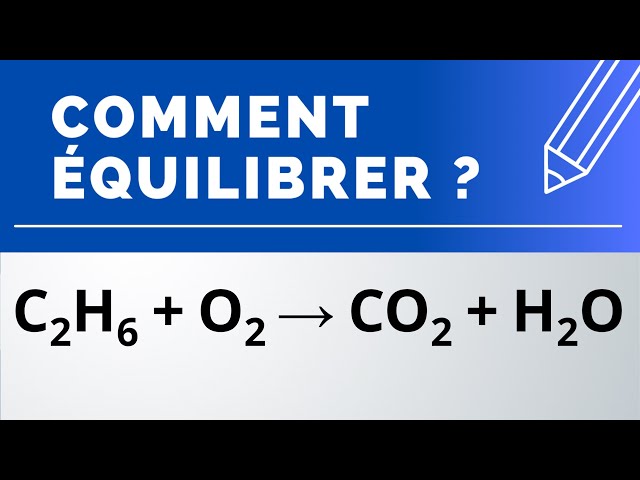Balance the following equation by oxidation number method. C6H6 + O2 → CO2 + H2 O - Sarthaks eConnect | Largest Online Education Community

SOLVED: Balance the equation by inserting coefficients as needed: equation: C6H6 + O2 -> CO2 + H2O Classify the reaction: combustion
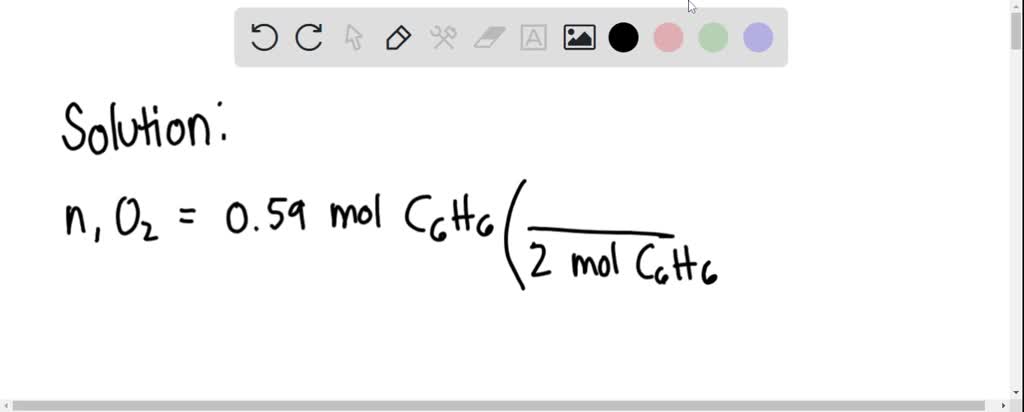
SOLVED: Benzene (C6H6) burns in air according to the following equation shown below. How many moles of O2 are required to react with 0.59 mol of C6H6? 2 C6H6(l) + 15 O2(g)

Balance this equation by oxidation number method (iv) C6H6 + O2 → CO2 + - Chemistry - Redox Reactions - 11918394 | Meritnation.com

C6H6+O2=CO2+H2O balance the chemical equation. c6h6+o2=co2+h2o benzene and oxygen reaction - YouTube
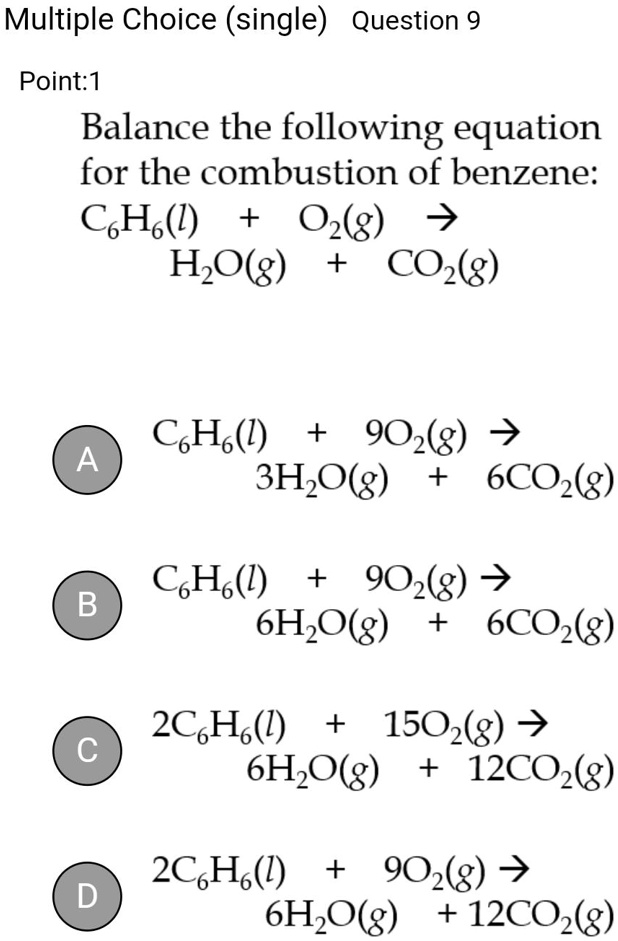
SOLVED: Multiple Choice (single) Question 9 Point: 1 Balance the following equation for the combustion of benzene: C6H6(l) + O2(g) â†' H2O(g) + CO2(g) C6H6(l) + 15O2(g) â†' 6H2O(g) + 6CO2(g) A

Drill #4B 2/10/15 Predict the products and balance the equation for the following double-replacement reaction: Na2CrO4 (aq) + Ba(OH)2 (aq) - ppt download
how to balance chemical equation c6h6+o2 arrow mark co2+h20 but. i tried and in RHS why should we put 12 next to carbon instead of that we can put put 3 so
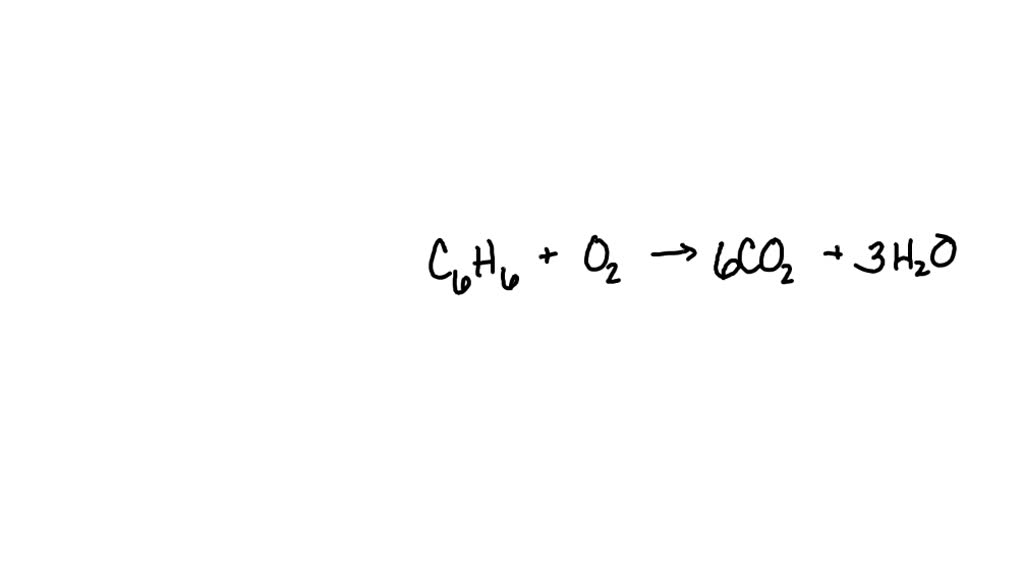
SOLVED: What is the coefficient value of CO2 when the following equation is completely balanced? C6H6 + O2 ————> CO2 + H2O